Transferrin Test Kit
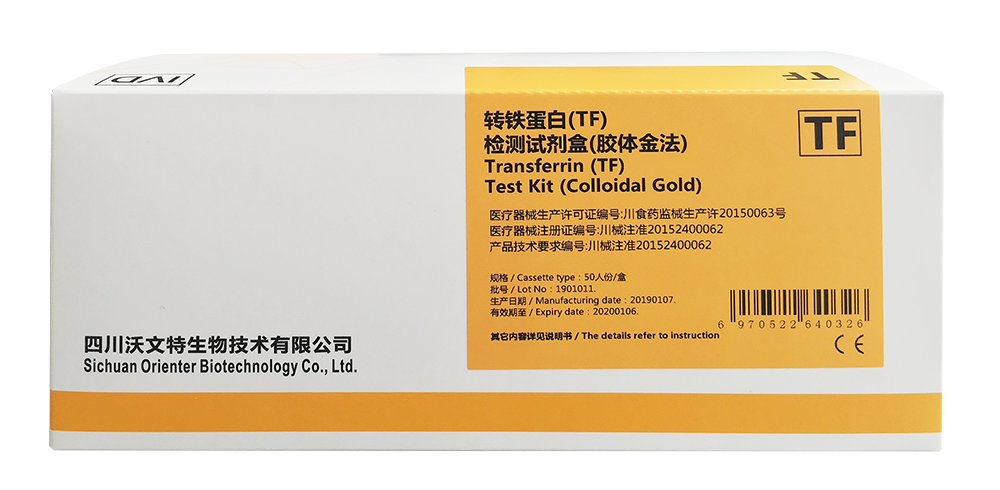
【Product Name】
Transferrin (TF) Test Kit (Colloidal Gold)
【Packaging Specifications】
Cassette type: 50 pcs/box.
【Storage conditions and validity】
Storage conditions: Store at 4 ~ 30 ℃ away from light and keep dry, shall not be frozen. Before using can open the aluminum foil packing, after opening should be used within 1 hour.
Validity: 12 months.
【Suitable Instrument】
This test kit applies to Fully Automatic Digital Feces Analyzer of Sichuan Orienter Biotechnology Co., Ltd. (Model: WWT/FA160, Registration No.: No. 2410087 by Sichuan food and drug administration)
Machine Testing (Apply to WWT/FA160 Fully Automatic Digital Feces Analyzer.)
1 Place the sampled tube on the tube rack, tear off the aluminum foil bag to take out the test kit and place it in the designated position of the instrument.
2 The instrument automatically sampling, mixing, adding, collecting the image of test results and auxiliary interpretation, the operator can also interpret the results based on the acquired image.
3 The operation of instrument refers to the instruction manual of WWT/FA160 Fully Automatic Digital Feces Analyzer.
【Positive judgment】
Transferrin content greater than or equal to 40ng/ml, test result is positive.
Refer to the confirmatory test of the critical value at home and abroad, the percentile method of unilateral 95% to determine the critical value, I.e., when the analyte at this level is repeatedly measured, the lowest concentration of positive result detected at 95% confidence level or more, is taken as positive judgment value.
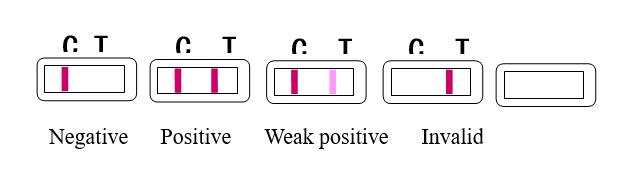
【Test Results】
Cassette type
【Matters Needing Attention】
1. The kit is suitable for the detection of stool sample for in vitro diagnosis.
2. Due to the technical and operational errors may occur, but also due to the existence of interfering substances in the sample, the results may be wrong. The suspicious results should be further detected, such as endoscopy.
3. TF test kit (colloidal gold) for one-time use, please use within the validity period, must not be reused. If the kit stored in refrigerators, freezers and other low temperature environment, recommend use after the indoor temperature balancing.
4. In the operation process, please abide by the laboratory safety operation specification, and the biological protection should be made. Stool samples and all used items should be properly handled according to the relevant laws and regulations.